Definition Of Extensive Property In Chemistry
An intensive property is a property of matter that depends only on the type of matter in a sample and not on the amount.
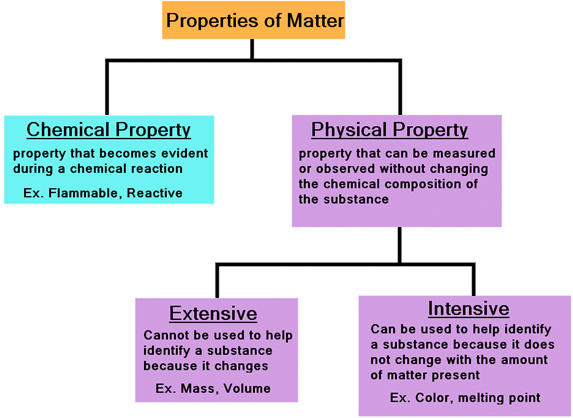
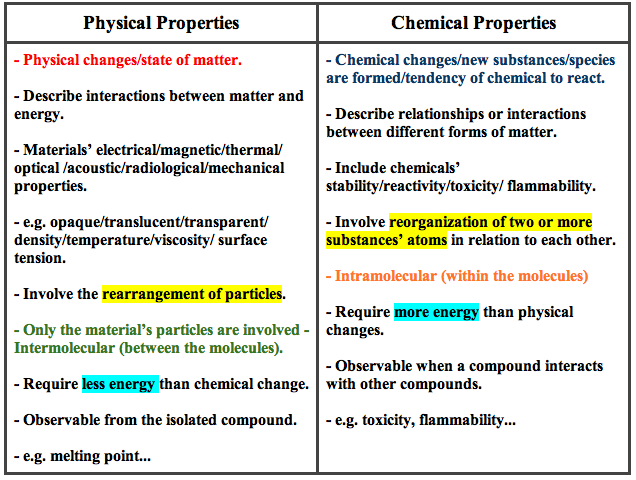
Definition of extensive property in chemistry. Mass and volume are examples of extensive properties. All properties of matter are either extensive or intensive and either physical or chemical. Like other physical properties an extensive property may be observed and measured without any chemical change reaction occurring.
Physical and chemical properties of matter. 1 a property that depends upon the amount of material in a sample. However it can become an intensive property if it is considered as a unit value such as the molar volume the volume of a mole of the substance.
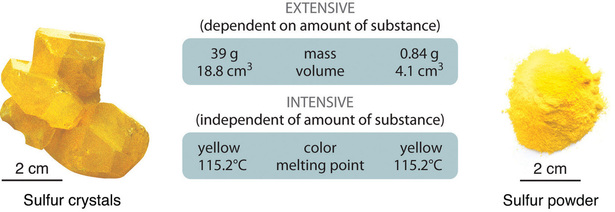
For example volume is an extensive property. Extensive property is a property that changes when the amount of matter in a sample changes. Color temperature and solubility are examples of intensive properties.
An extensive property is a property that depends on the amount of matter in a sample. Examples of extensive property in the following topics. An extensive property is a property of matter that changes as the amount of matter changes.
There are some extensive properties that can be used as intensive. An intensive property is a property of matter that depends only on the type of matter in a sample and not on the amount. Examples are mass volume length and charge.
2 any property that depends on the size of the system m v. Conversely intensive properties do not have any dependence on the amount of the material in the system the intensive properties of a substance will not change regardless of how much there is. Both extensive and intensive properties are physical properties which means they can be.
Mass and volume are examples of extensive properties.